Structure of the Chaperonin Protein, Simplified
Structure of the Chaperonin Protein, Simplified
Submitted by Oksana Sergeeva and Jonathan King in the King Lab at MIT
MIT Department of Biology
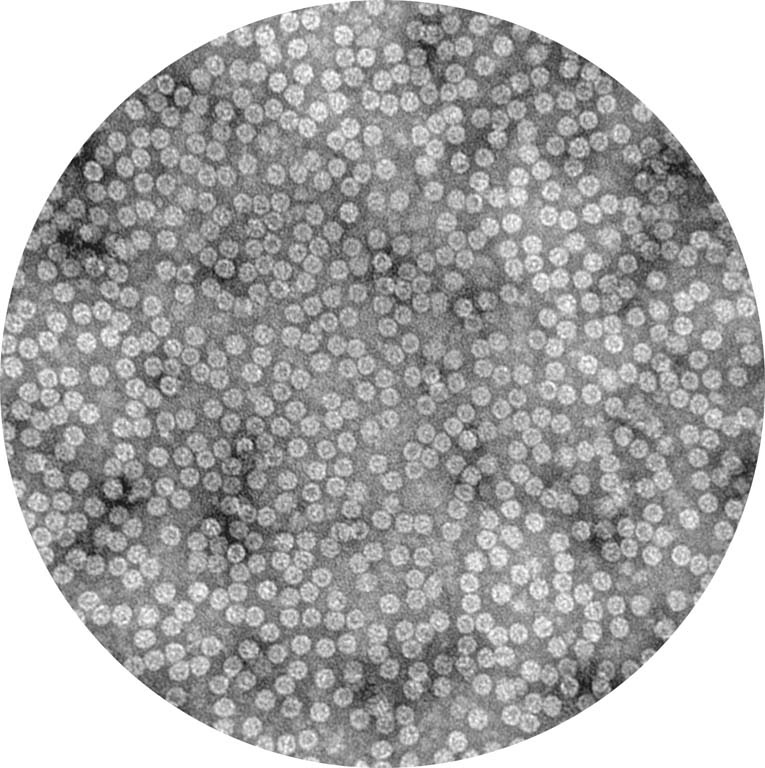
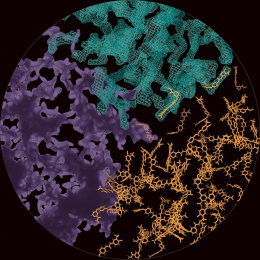
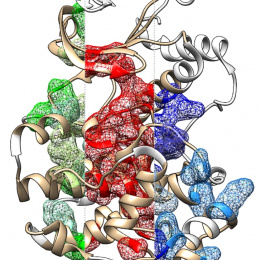
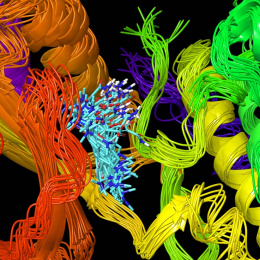
This is the structure of a protein called a chaperonin that folds other proteins by encapsulating them into an inner cavity and then later spitting them out. This particular chaperonin is engineered to have only one subunit rather than the eight that make up the native chaperonin found in all human cells. When we were studying this particular subunit, we found that it can take on the structure and function of the complete complex, giving us a more simplified system to work with to understand chaperonin function. The native chaperonin is essential for the cell to live.